Neural Circuits and Behaviour
We have established that the Hox transcription factors Deformed (Dfd) is critically required for feeding behavior, importantly
for the establishment of the stereotypic feeding movements.
Movie of a Drosophila larva extensively moving its head with the associated mouth hooks, which are required for hatching
and feeding.
Using genetic, molecular, genomic and behavioral approaches we have demonstrated that Dfd is required throughout the formation
of the feeding unit by directly controlling neuronal specification, axon outgrowth, synapse formation and
genes with roles in neurotransmission. Importantly we have found the expression of the Dfd target Ankyrin2-XL
to be reduced and synapse architecture to be changed after late interference with Dfd activity, demonstrating
that the microtubule-organizing complex required for synaptic stability and connectivity is under direct
Hox control.

Muscle bundles (red) associated with different parts of the feeding apparatus are innervated by Dfd positive motoneurons
(green) to allow synaptic transmission and feeding movements. Functional synapses are labelled with DVGlut
(blue).
We now use this system to study on the one hand how Hox TFs control the communication and interaction of motoneurons with
their target muscles, which is critical for the establishment of region-specific motor units and thus for
coordinated movements. On the other hand, we aim to understand how Hox TFs execute their neuro-protective
function we have uncovered, which is relevant in the context neuro-degenerative diseases and ageing.
Stem Cell Control
Animal tissues and organs are generated and maintained by stem cells, which fulfil this task by keeping an undifferentiated
state, while at the same time generating daughter cells that are lineage-restricted and undergo terminal
differentiation. Due to this unique ability stem cells supply new cells to replace dying, lost or damaged
ones in adult animals and consequently are critical for tissue homeostasis. However the life-long capacity
of a cell to divide also harbours dangers, since defects in the control of this process can result in tissue
degeneration or tumorigenesis.

Schematic drawing of a Drosophila larval testis with the the testis niche, the hub, the germline stem cells (GSCs), somatic
cyst stem cells (CySCs) and the GSC progenies, the gonioblasts (GBs) highlighted (image by Stephanie
Guse).
Pioneer studies gave insights into how local microenvironments, or niches, control the maintenance and activity of stem cells.
We use the Drosophila testis to study stem cell behaviour and control and have previously
shown that the Hox protein Abdominal-B (Abd-B), through local signaling between adjacent cell types, provides
positional cues for integrin localization, which critical for the placement of the distant stem cell niche
and stem cell activity. Now we use genome-wide approaches and the Hox TF Abdominal-A (Abd-A) and have identified
many novel regulators of stem cell behavior in the Drosophila testis.
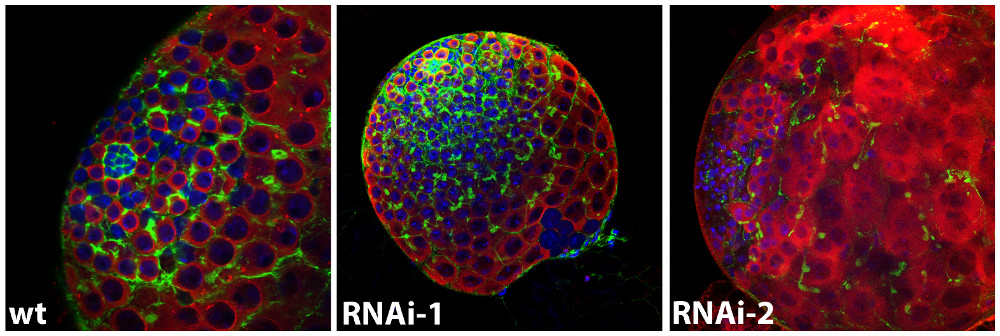
Images of a Drosophila wild-type larval testis in comparison to testes, in which the activity of two newly identified stem
cell regulators is knocked down. The germline lineage is shown in red (Vasa), the somatic population
in green (actin), the nuclei are highlighted in blue (DAPI).
Activity and Specificity
How Hox proteins acquire spatio-temporal precision and cell-type specificity in regulating their target genes despite their
broad expression is a fundamental question in the field. Hox proteins seem to solve this problem by interacting
with different cis-regulatory modules and diverse sets of of transcriptional partners to form large regulatory
complexes thereby enabling Hox transcription factors to achieve their highly diverse and specific transcriptomic
outputs. We use the Isolation of Nuclei TAgged in specific Cell Types (INTACT) method to dissect the cis-regulatory
requirements for tissue- and stage-specific Hox function by identifying in the embryonic mesoderm and nervous
system chromatin modifications, binding profiles (ChIPseq) and direct target genes (RNAseq) for different
Hox proteins. In parallel, we have established proteomics approaches to isolate tissue- and stage-specific
Hox interaction partners, the so-called Hoxosomes. Our genomic and proteomic findings are tested on selected
enhancers in vivo.

Metabolism and Proliferation
Tissue homeostasis in stem cell compartments is controlled by developmental pathways, environmental stimuli and the metabolic
state. While there is a robust knowledge about the developmental genetic programs, rather little is known
about how these are coordinated with metabolic signals. We elucidate the mechanisms of how metabolic signatures
are sensed and translated into developmental decisions using the Drosophila eye
epithelial tissue as a model system.

The image shows a Drosophila eye-antennal disc, where cells are labelled by eyeless-FLP induced GFP expression (green). Proliferating
cells are highlighted by phospho-H3 staining (magenta) and differentiating photoreceptors located posterior
to the morphogenetic furrow are labelled with ELAV (red).
Starting points for the project are candidate genes with metabolic function recently identified in large-scale genomic screens,
which have strong genetic interactions with developmental regulators, in particular the Notch signaling pathway.
We study this interaction at the genetic, genomic and metabolic level making use of genetically encoded nanosensors
and metabolite analysis. Together, this study will not only shed light on how important developmental signaling
pathways are interlinked with metabolic processes but also how disturbance of these intersections can cause
aberrant cellular behavior resulting in severe diseases in man.

Adult fly eyes showing how the intersection of pathway activation and disturbance of the metabolic properties of cells affects
proliferation and tissue size.
